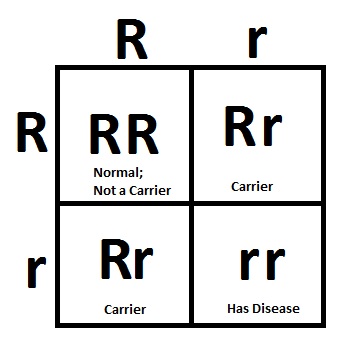
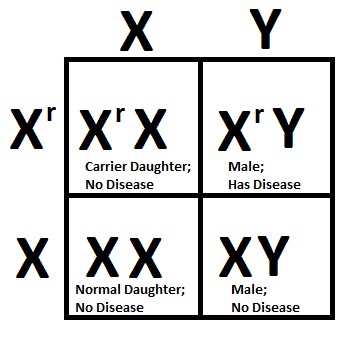
To get an idea of how gene regulation and expression work in your cells, imagine someone went through your house and turned on all your electrical components at full-blast. The radio and the TV would be blaring, the fridge would turn into a freezer, and your home heating system would melt you into a puddle of sweat. It’d be chaos!
To get things back to normal, you’d need to go through and turn off the things you didn’t need on or adjust the volume and settings so you’re getting the proper amount of sound, heating, cooling, etc. Your home is a precisely-tuned system when you think about it!
Your cells work the same way. Each one of your 37.2 trillion cells has a complete copy of all of the genes within your DNA, but they’re not all turned on at the same time. The cells that make your toenail, for example, still have the genes that a heart muscle cell or a bone cell uses to make heart and bone-specific proteins. So what distinguishes the toenail-making cell from the bone-making cell and the heart muscle-making cell if they all have the same genes?
It’s all in how each cell expresses them! The toenail-making cell, for example, does have the gene to make myoglobin (a protein found in muscle cell), but it has disabled the gene for that particular protein (could you imagine if it did? Your toenails would turn red and could absorb oxygen!).
Instead, the toenail-making cell creates keratin (a protein found in your nails), while the heart muscle-making cell disables this gene.
How do they do it?
Each cell in your body has ways to turn genes on and off, just like you going through your house and flipping the on/off switches. Cells can also adjust the “volume” of each gene and control how many proteins are produced.
This process is called gene regulation and expression, and there are many ways that cells can do it. In fact, we haven’t even discovered them all yet! In this article, we’ll go over some of the most common ways that cells can regulate and express genes.
The Big Picture: What’s happening in the cell, and where can we control it?
There are a lot of steps that happen in the process that starts with a cell’s DNA and ends with a protein. Genes can be regulated at each one of these steps, so it’ll be helpful to go over them what they are.
There are two major cellular processes that produce proteins: transcription and translation. In transcription, the goal is to produce an mRNA (messenger RNA) copy of an entire gene from its DNA template. This mRNA template is then translated into an actual protein.
First, DNA is transcribed into an mRNA template when RNA polymerase (the gene copying machine) attaches to a gene on the DNA strand. The RNA polymerase slides down the DNA and spits out and exact mRNA copy of the gene.
Our little mRNA template then floats peacefully out of the nucleus and into the cytoplasm (the rest of the liquidy goop that makes up the inside of the cell), where a ribosome attaches to it. The ribosome then slides down the mRNA template, reads the code, and assembles a chain of amino acids based on whatever the code says. At the end, the ribosome falls off and the chain is finished—except now the amino acid chain is called a protein.
Transcription: The First Regulation Point
Transcription is the start of everything, so it’s easiest to regulate which genes are used or not at this point. That way, your cell isn’t wasting time with stuff it’s already built further down the road.
It’s a bit like cancelling an unwanted order before it’s shipped to you. Otherwise, you’d have to wait for it to get to you and then decide you don’t need it anymore. Your time and money will have been wasted.
Similarly, if you need to boost production of a certain protein, it’s also fairly easy to regulate at this point as well. If you need more proteins, just place an order for more!
DNA Methylation
There are several ways that the cell manages to up-regulate or down-regulate its mRNA levels. Perhaps one of the coolest is by turning particular genes off through a process called DNA methylation.
Each gene is outlined by two separate DNA sequences in the genome—a promoter sequence (the start of the gene), and a terminator sequence (the end of the gene). They are like the front and back cover of a book; they serve to denote the beginning and the end of the information contained within.
There is also a molecule called a methyl group, which is made up of one carbon atom and three hydrogen atoms. This methyl group can bind to lots of things—it’s kind of like a sticky note! When a methyl group binds to a promoter sequence of a particular gene, it makes it virtually “invisible” to the RNA polymerase. If it can’t find the promoter sequence of the gene anymore, it can’t start translation. It masks the gene and turns it off!
One of the really cool things about DNA methylation is that these methyl groups are even reproduced along with the DNA in DNA replication. For example, if a metabolism gene is methylated (turned off) and that cell splits in two, the new DNA will also have a methyl tag on that same gene.
This can even apply to genes that are passed down when you reproduce. Based on how you live and which genes your body turns on or off, your kids might inherit genes that are physically present, but not turned on!
Enhancer Sequences
What if you need to turn up the volume for a particular protein? That’s where enhancer sequences come in. They work just like promoter sequences in a gene, except that if a particular protein binds to an enhancer sequence, it ramps up mRNA production even further. It’s as if there was a giant neon sign erected over the gene that says, “Hey, RNA polymerase! Come transcribe this gene! It’s really important!”
Translation: The Second Regulation Point
mRNA transcripts are pretty powerful little pieces of code. They can be read many times per minute, and each transcript has a half-life of about seven hours. That means every seven hours, the number of mRNA transcripts made at a certain time point will be cut in half. When you look at it that way, one single mRNA transcript can be used to make hundreds of proteins!
Seven hours is a long time to be hanging out around the cell. Sometimes, the cell needs to adjust gene regulation right away—not seven hours from now. It doesn’t have time to wait for those mRNA transcripts to dissipate on their own.
For example, let’s say you’re going for a hike and you surprise a bear in the bushes. You’ll get a sudden jolt of adrenaline (and hopefully you don’t mess your pants!), and your cells will instantly try to boost metabolism-related genes so that they can burn energy faster and be prepared for the flight-or-fight response. To do that, it needs to get all of the genes related to storing fat out of the way (now’s no time to store fats—you need to burn them for energy!).
One of the ways the cell can do this is by boosting production of proteins that break down mRNA strands faster. That way, the ribosomes will be unoccupied and free to make more of the metabolism-related proteins to burn fuel faster and get you out of there.
Post-Translational Modification: How To Alter What’s Already Been Made
If the cells in your body need to change their function, they may also need to change the proteins that are already floating around in the cell. There are two main ways that the cell can do this:
Protein Phosphorylation
Some proteins come with a built-in on/off switch in the form of a phosphate group—a molecule composed of one phosphorus atom bound with four oxygen atoms. It works just like the DNA methyl groups: if a phosphate group is added onto a protein, it can change the function of the protein.
Each protein reacts differently to being phosphorylated. Sometimes the phosphate group can turn the protein on, and with other proteins, the phosphate group turns the protein off.
Protein phosphorylation is especially important for many metabolic processes. For example, hormone-sensitive lipase is a protein that is turned on when phosphorylated. It breaks down fat stored in your cells so that it can be used as fuel when you exercise.
Glycogen synthase, on the other hand, is a protein that creates glycogen to be used as a short-term energy source. It turns off when phosphorylated.
Ubiquitin
What if a protein is no longer needed at all? You would need to find some way to go through and mark each individual protein for removal, similar to foresters marking certain trees for removal followed by a team to harvest these marked trees.
The cell does this on a microscopic level. If a cell knows that it’ll no longer need a certain protein, it’ll attach another protein—ubiquitin—to it. Once a protein is “tagged” with ubiquitin, it’s marked for removal.
Ubiquitin-marked proteins make their way to an area of the cell called the proteasome, which is like a cellular recycling center. When a protein goes into the proteasome, it’s chopped up into its component parts—amino acids. These free amino acids float out of the proteasome and can be reused in another protein.
Why study gene regulation and expression?
If we understand how genes are expressed and regulated in organisms, we might be able to manipulate them to make people (or other organisms) healthier. It’s a lot of work to put new genes into an organism. If we could just use the genes that are already there, it would be far easier!
For example, people’s metabolism changes as they get older. Usually people are thin and athletic when young, but as they age they begin putting on more weight. When you’re older, those genes that kept you slim and trim when young are still there, they’re just turned off.
But, what if we could turn those genes back on again? We could make people slim again just like when they were younger!
So far, we only know of a handful of ways that genes can be regulated and expressed, but it’s very likely that there are many more. Who knows—maybe you can be the one to discover a new regulation or expression method, or crack the code in how to turn individual genes on or off!